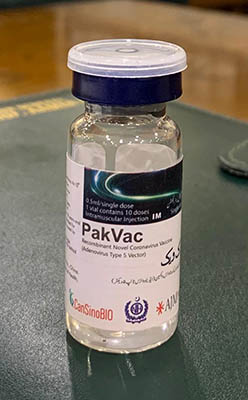
Locally finished vaccine was produced with help of China’s CanSino and is aimed at ensuring adequate supply for inoculation drive
Pakistan on Monday introduced the PakVac vaccine against coronavirus, manufactured locally using raw materials provided by China’s CanSino Bio Inc.
“Congratulations to the National Institute of Health team and its leadership for successful fill/finish (from concentrate) of the CanSino vaccine with the help of CansinoBio Inc. China,” Special Assistant to the P.M. on Health Dr. Faisal Sultan announced in a posting on Twitter. “The product has passed rigorous internal QA [Quality Assurance] testing. An important step to help in our vaccine supply line,” he added.
The raw materials for the vaccine were airlifted from China to Islamabad last month when officials of the NIH had said they would commence local production and launch distribution from June. An official of the Ministry of National Health Services has said that due to the agreement on technology transfer, it would be able to produce three million doses per month.
The PakVac vaccine utilizes the CanSinoBio single-dose vaccine; Pakistan was among the countries that were part of the Phase III clinical trials for it. According to Dr. Sultan, it has been proven to have 75 percent efficacy at preventing symptomatic cases of the coronavirus in Pakistan.
The PakVac vaccine is believed to significantly reduce Pakistan’s dependence on other countries for COVID-19 vaccines. The country, which launched its vaccination drive in February, has suffered delays due to promised doses under the COVAX regime being delayed as a result of neighboring India halting exports in light of the devastating second wave of the pandemic sweeping through it.
Separately, the University of Health Sciences has announced that it has started working on testing a single-dose nasal COVID-19 vaccine that would be administered through a spray and would not require a syringe. The varsity hopes to launch clinical trials for the vaccine in six to eight weeks after securing all relevant approvals.
The vaccine would be capable of generating an immune response at the site of infection and within the respiratory passage.
